Published on 01/06/2020
As a child, Christina Fasser could ski, but she struggled with tennis. She also fumbled to find things in the dark. “I just thought I’m clumsy,” she says. “I’m not as good as the others.”
Then came the news. At age 13, Fasser was diagnosed with retinitis pigmentosa, a degenerative eye disease that damages retina cells. For Fasser, the disease explained a lot. She wasn’t clumsy. Rather, she was night-blind and lacked the 3D vision needed to play ball sports. “If you are born with a restricted visual field, you have no idea what it means to see fully,” she says.
There are no effective therapies available for Fasser’s retinitis pigmentosa. The same holds true for patients with other inherited retinal disorders that degenerate the light-sensitive rods and cones of the retina, such as Leber’s congenital amaurosis and Usher Syndrome, though clinical trials are ongoing. Approximately 2 million people worldwide suffer from these diseases and the life-limiting disabilities that accompany them, according to research from the Institut de la Vision in Paris.
A vision hacker of sorts
Yet new hope is coming through research, particularly that of Botond Roska, a neurobiologist at the Friedrich Miescher Institute for Bio-medical Research (FMI) in Basel, a research center affiliated with the Novartis Institutes for BioMedical Research (NIBR) and with the University of Basel.
In the mid-1990s, Roska emerged from medical school feeling uneasy. Being a doctor would sometimes require him to offer his patients therapies that he couldn’t explain. “That’s not what I wanted to do,” he says. “I wanted to understand.”
So Roska went back to school. He had studied mathematics before medical school, and the eye offered a possible way to combine the two fields. At the University of California, Berkeley, he began what has turned into a decades-long quest to understand the retina. “Botond asks questions with a totally unbiased mind. He has an impressive mastery of the field, but he is also very interested in driving that knowledge forward to benefit patients,” says ophthalmologist José-Alain Sahel, director of the Institut de la Vision, and one of Roska’s longtime collaborators.
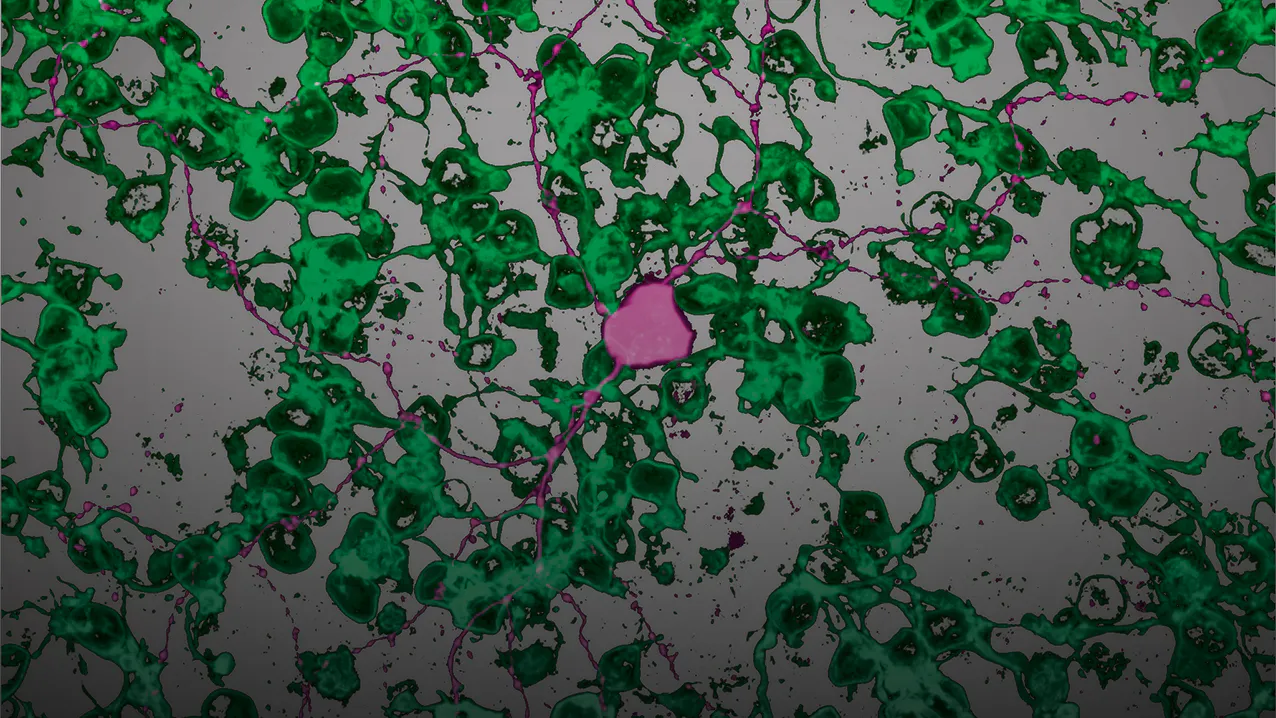
Roska found that the retina, a slip of light-sensitive tissue lining the eye, is actually an astonishingly complex biological computer. As a result, he has become a vision hacker of sorts, using the tools of neuroscience and genetics to reverse-engineer the retina to understand how it functions and how disease-related damage might be repaired.
Turning the switch on
In 2005, he started in his own laboratory at the FMI. FMI investigators focus on the fundamental science Roska believes is essential for progress. “You really have to dissect the different cell types and computations,” says Roska. “With that understanding, you get insights into potential therapies.”
The retina, transparent and just half a millimeter thick, contains 10 layers of cells that form a complex signal processor. This biological computer is coated with over 100 million photoreceptive rod and cone cells, the outer tips of which sense light and make sight possible. Damage to these tips, which occurs in genetic diseases such as retinitis pigmentosa, causes blindness.
It turns out, however, that a molecular switch controls the growth of the light-sensitive outer segments of cone cells. This switch consists of two microRNAs, which are small molecules that silence genes. Botond Roska published his discovery of the switch, in Neuron in 2014. The microRNAs themselves may have therapeutic potential, says Roska, “but before that there is something simpler, which is to use them to study disease mechanisms.”
Roska’s lab uses mice to model blindness diseases, but mouse retinas and human retinas are quite different. For instance, mouse retinas lack a fovea, a tiny, conedense region of the human retina that allows for high-resolution color vision. Also, some genetic diseases that cause blindness in humans do not progress the same way in mice.
“We need human models,” says Roska. The first human retina-in-a-dish, created using human stem cells by a team in Japan in 2012, was a pivotal advancement. But these lab-grown retinas could not sense light. So Roska decided to add his newly discovered microRNA switch to a growing retina-in-a-dish. The cone cells began changing. Their outer segments started to grow and function and the cells showed activity in response to light.
“We thought this is very nice,” says Roska. “Now we can develop a disease model for many human genetic retina diseases in a dish.”
A living model to learn from
Roska’s recent efforts have been focused on building retina-in-a-dish models from the cells of real patients. He and collaborators in Switzerland begin by taking a skin sample from a person with a gene mutation associated with a disease such as retinitis pigmentosa. With the right biochemical cues, these skin cells are coaxed back into stem cells, called induced pluripotent stem (iPS) cells, that become the building blocks of the model retinas-in-a-dish.
Additional cues, including modulation of Roska’s microRNAs, prod the iPS cells to transform into different types of retina cells, and to self-assemble into a living, light-sensing model retina.
“These lab-grown retinas are obviously not an eye, but they have a lot of the rudiments of an eye, so we can use them to learn a lot,” says Tewis Bouwmeester, Head of the Developmental and Molecular Pathways department at NIBR in Basel, who is collaborating with Roska to build new models of blindness diseases. The project supports NIBR’s exploration of molecular signaling pathways that drive organ development – and the misfires that cause disease – to gain insights which could lead to new potential therapies, in this case for inherited causes of blindness.
Roska hopes that these retina-in-a-dish disease models will begin to show symptoms. “The treatment will come when we can clearly see the disease in a dish and learn how to treat it,” he says.
FMI and NIBR in close cooperation
This dual effort to understand and cure is emblematic of Roska’s work. “He is absolutely dedicated to fundamental discovery, but he is also interested in turning that understanding into ways to treat blindness diseases,” says Bouwmeester.
Bouwmeester is also collaborating with Roska on a simpler way to model blindness diseases in rods and cones grown without the rest of the retina. Creating a lab-grown retina is time-consuming, so their goal is to build cultured cell models that can be produced quickly and efficiently in large volumes.
If the work is successful, these cell models could be used to screen potential therapies to see which works best to repair the damage caused by disease. From there, promising drug candidates can be tested in the more realistic retina-in-a-dish model.
Look to the future
Meanwhile, for Fasser, Roska’s progress translates into hope. “His work has opened up completely new ways to look at restoration of vision,” she says.
Now totally blind, Fasser relies on modern technologies to function. She uses text-to-speech programs for reading, light detectors to light her home for visitors, and color sensors to choose clothing and sort the wash. But, as president of the advocacy group Retina International for over two decades, she has a loftier goal in mind. “To find a cure,” she says.
You may also be interested in the following:
Thanks for reading Live.Magazine
Stay connected by subscribing to our newsletter
SubscribeBy submitting your email, you consent to Novartis AG collecting and processing your email data for Novartis internal use, in accordance with our privacy policy, and by protected technical means.